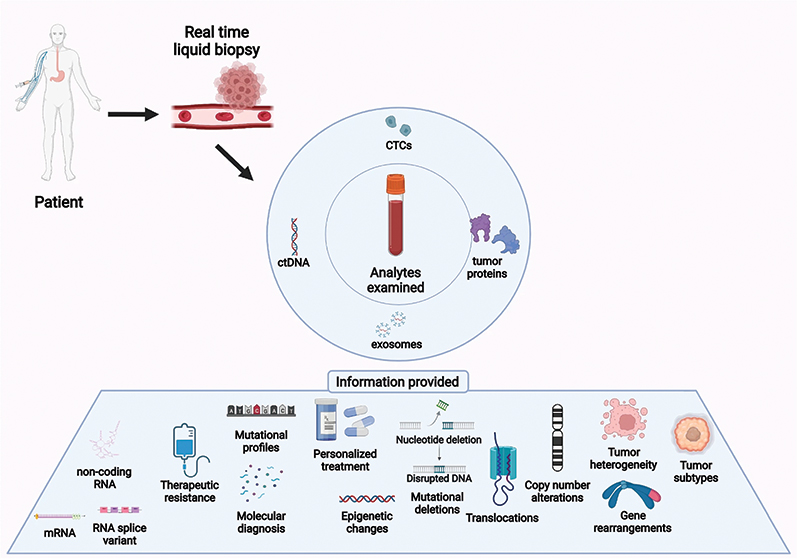
Figure from: Liquid Biopsy, Lone SN, et al. Molecular Cancer 21, 79 (2022). Used under the Creative Commons license: creativecommons.org/licenses/by/4.0/
What Is Liquid Biopsy and How Can It Help Our Patients?
Liquid biopsy is a relatively new diagnostic modality that uses blood, urine, or other biological fluids to detect cells, nucleic acids, or other cellular components.
Oncologists can use this tool to detect circulating tumor cells (CTCs), cell-free DNA (cfDNA), and exosomes. Circulating tumor cells are intact cells that have separated from the primary tumor and are found in circulating blood. cfDNA is degraded DNA fragments that are released into the blood as a result of apoptosis or necrosis. Exosomes are vesicles secreted by cells to allow cellular communication and to transport molecules such as proteins.
What Could Liquid Biopsy Tell You?
Liquid biopsy may serve as a prognostic indicator for some cancers. The presence of cfDNA or CTCs may be associated with increased tumor burden, aggressiveness, and decreased time to relapse. It may also be used to help monitor response to treatment and be an early marker for tumor recurrence or resistance to treatment. In humans, liquid biopsy can help guide targeted or personalized therapies. There are many benefits as well as limitations to liquid biopsy (see table below).
| Benefits | Limitations |
|---|---|
| Earlier diagnosis | Concentrations in blood are very low, requires specialized techniques and 7-9 mls of blood |
| Less invasive | Less effective for early and localized disease |
| Less costly (depending on local market) | False positives |
| Easily repeated/can be used for monitoring | Not sensitive if more than one tumor type present |
| Can assess tumor heterogeneity | Does not give information about tumor microenvironment or architecture |
| Good for tumors that cannot be biopsied | Targeting therapies are lacking in veterinary medicine |
Should You Incorporate This Tool into Your Practice?
Liquid biopsy has recently become available in veterinary medicine. The most widely available test is OncoK9 liquid biopsy from PetDx. This test is only available at Idexx and Antech. It is not offered at the University of Illinois Veterinary Diagnostic Laboratory.
Our Cancer Care Clinic does not currently use this test because it is still early in development. The OncoK9 screening test requires a test kit provided from the company or lab (available at Idexx and Antech).
The test has primarily been marketed to general practitioners as a screening tool for senior dogs or high-risk breeds, with the goal of early cancer detection to extend the pet’s survival. It is most sensitive for the detection of lymphoma, hemangiosarcoma, and osteosarcoma, all of which are usually diagnosed through standard procedures. Other cancer types can be detected, though these can vary.
The test has demonstrated sensitivity of 85.4% across the three cancers listed above, which are among the most aggressive canine cancers. The test has a specificity of 98.5%, which corresponds to a false positive rate of just 1.5%.
While the test is sometimes less costly than a biopsy with histopathology or imaging, the annual cost for screening should be considered. If used as a screening test and cancer signal is detected, practitioners should be ready to discuss what to do with the results and what it could mean for the patient.
The next step would be full staging (lab work, 3 view chest radiographs, and abdominal ultrasound/aspirates) to find and diagnose the tumor, if present. If no tumor is detected after these additional tests, the company recommends a full body CT scan for an otherwise healthy patient in which a cancer signal was detected.
Molecular tests such as these should not be used alone, rather to complement a good physical exam, history, owner’s observations, routine diagnostics, and consultation with specialists. Liquid biopsy may be one piece of the diagnostic puzzle, and having more pieces of information may provide a more accurate picture.
It remains unclear how liquid biopsy will fit into cancer detection in general practice. It may come to serve a useful purpose in confirming an elusive diagnosis or monitoring tumor recurrence after treatment. Research is currently ongoing to evaluate these and other uses.
References
• Lone SN, Nisar S, Masoodi T, et al. Liquid biopsy: a step closer to transform diagnosis, prognosis and future of cancer treatments. Mol Cancer 21, 79 (2022).
• Palmirotta R, Lovero D, Cafforio P, et al. Liquid biopsy of cancer: a multimodal diagnostic tool in clinical oncology. Ther Adv Med Oncol. 2018.
• Flory A, Kruglyak KM, Tynan JA, McLennan LM, Rafalko JM, Fiaux PC, et al. (2022) Clinical validation of a next-generation sequencing based multi-cancer early detection “liquid biopsy” blood test in over 1,000 dogs using an independent testing set: The CANcer Detection in Dogs (CANDiD) study. PLoS ONE 17(4): e0266623.
By Joanna Schmit, DVM, MS, DACVIM (Oncology)