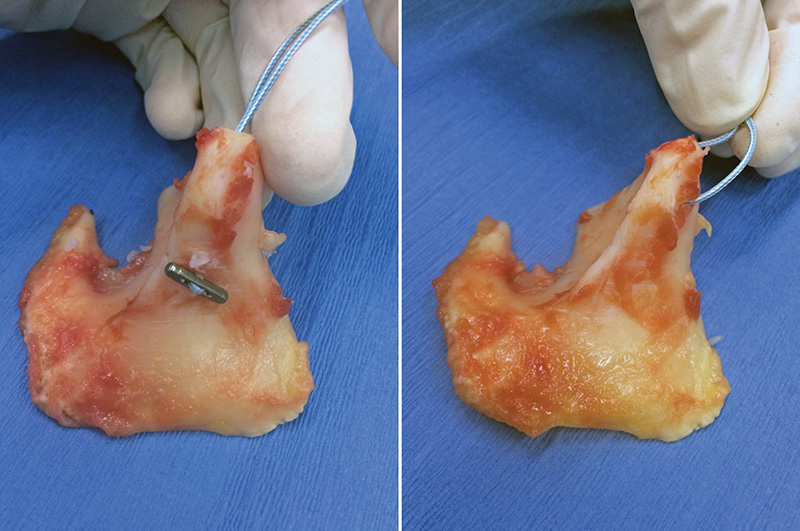
A titanium suture button is used to “toggle” the suture material (light blue) to the body and muscular process of the arytenoid cartilage (left). Note the difference with the picture on the right where the suture is only anchored to the muscular process.
By Santiago D. Gutierrez-Nibeyro, DVM, MS, DACVS-LA, DACVSMR (Equine)

Recurrent laryngeal neuropathy (RLN) (formerly known as laryngeal hemiplegia) is an important cause of decreased performance in horses. The condition creates partial airway obstruction during exercise caused by the dynamic collapse of one of the arytenoid cartilages (typically the left).
Prosthetic laryngoplasty can restore normal airway function by eliminating the dynamic collapse of the arytenoid. However, most horses treated with the standard prosthetic laryngoplasty technique experience postoperative loss of arytenoid abduction, narrowed cross-sectional area of the rima glottidis, and recurrence of dynamic collapse of the arytenoid due to suture pull-through from the arytenoid cartilage.
Many affected horses are retired prematurely from racing, or do not reach their maximal athletic potential, resulting in unwanted racehorses that could otherwise continue to have successful careers.
Modified-Toggle Technique
My research group has developed and tested in the laboratory a modified prosthetic laryngoplasty technique that eliminates suture pull-through from the muscular process of the arytenoid cartilage. The modified-toggle prosthetic laryngoplasty technique uses a suture button to “toggle” the suture material to the base of the muscular process of the arytenoid cartilage.
This modification eliminated suture pull through from the muscular process of ex-vivo laryngoplasty constructs subjected to cyclic loading designed to mimic postoperative cyclical adductory forces applied to the laryngoplasty during swallowing in vivo (research published in the American Journal of Veterinary Research). Also, the modified toggle prosthetic laryngoplasty constructs were found to have comparable airway mechanics in an ex-vivo model (research published in the journal Veterinary Surgery).
While this previous work is promising, clinical application of the modified-toggle prosthetic laryngoplasty technique is mandatory to establish if the modified technique is clinically superior to the standard prosthetic laryngoplasty technique to treat RLN in racehorses. If the modified prosthetic laryngoplasty technique is effective in treating Standardbred racehorses with RLN, we expect that it would also be beneficial to treat other horse breeds affected by RLN, such as Thoroughbred, Quarter Horse, Warmblood, and draft horses (e.g., Belgian).
Trial to Enroll 30 Horses
In January my research group opened a clinical trial to assess the short- and long-term efficacy of standard and modified-toggle prosthetic laryngoplasty procedures for the treatment of left-sided RLN in racehorses. If the modified-toggle prosthetic laryngoplasty technique developed at the University of Illinois is clinically superior to the standard prosthetic laryngoplasty technique to treat left-sided RLN in racehorses, clinical use of the modified-toggle prosthetic laryngoplasty technique can be advocated worldwide.
Thirty horses will be randomly assigned to undergo a standard or modified-toggle prosthetic laryngoplasty procedure. Standardbred racehorses (2 to 5 years old) will be included if they have a history of upper airway noise, poor performance, and endoscopic evidence of grade 3 or 4 left-sided RLN. All horses that fulfill the inclusion criteria will be enrolled and housed in the University of Illinois Veterinary Teaching Hospital for one week after the surgery.
Owners will remain unaware of which prosthetic laryngoplasty procedure was performed for their horses unless complications required disclosure (unmasking). Owners will pay $1,000 for the surgery and hospitalization, and funding from a grant received will for the study will cover the rest of the expenses.
Individuals interested in this clinical trial should contact:
Dr. Adriane Rhodes or Dr. Santiago Gutierrez-Nibeyro,
Large Animal Clinic:
(217) 333-2001 for referring veterinarians
or (217) 333-2000 for clients