OUR RESEARCH
Phospholipids and cholesterol are major constituents of biological membranes. Phospholipid bilayers fulfill important structural functions by segregating cellular contents from the surrounding environment, forming subcellular organelles and providing platforms for a variety of cellular processes. The fatty acyl moieties of membrane phospholipids exhibit considerable diversity in chain length and degree of saturation. These two parameters determine the biophysical properties of cell membranes, including their fluidity, curvature, and subdomain architecture. These factors in turn influence membrane-associated cellular processes, such as vesical trafficking, signal transduction, and molecular transport.
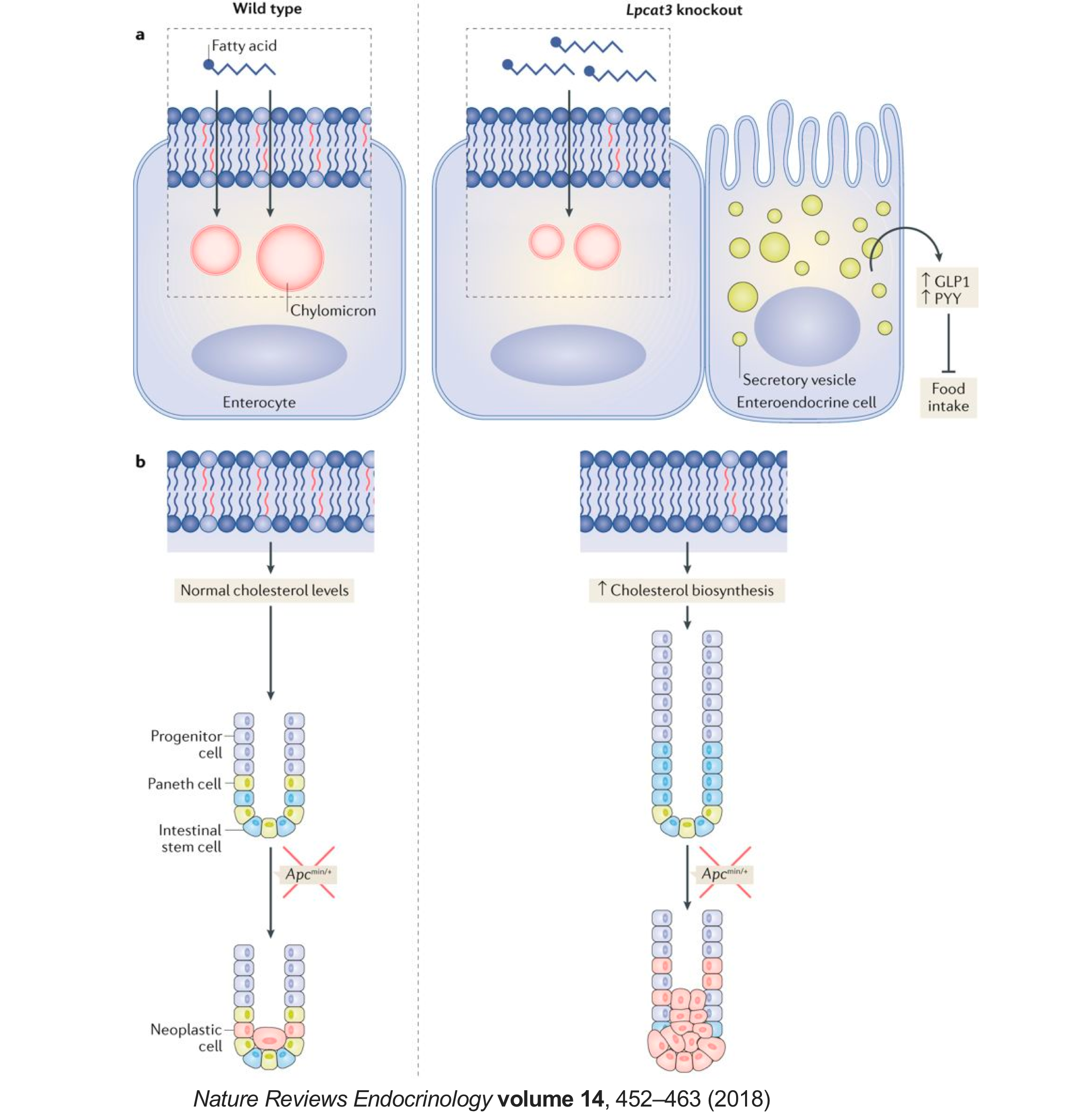
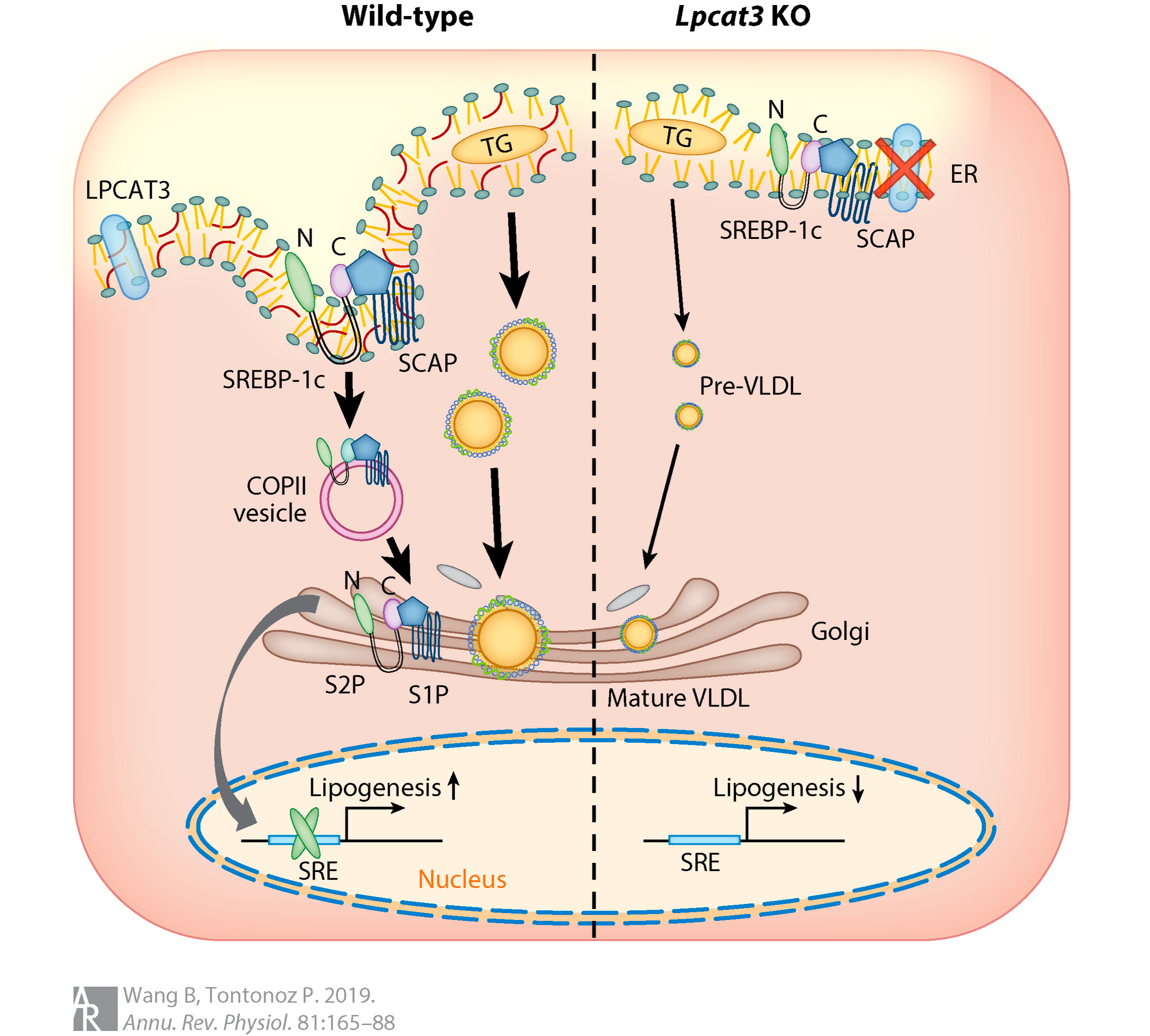
Our previous research has identified lysophosphatidylcholine acyltransferase 3 (Lpcat3) as a critical determinant of membrane phospholipid composition and biophysical properties. Lpcat3 catalyzes the incorporation of polyunsaturated fatty acids at the sn-2 site of lysophospholipids, giving rise to polyunsaturated phospholipids. The ability to manipulate membrane phospholipid composition in vivo by regulating Lpcat3 expression provides an unprecedented opportunity to study the consequences of changing the membrane composition in homeostasis and the pathogenesis of diseases in living animals. Using Lpcat3 knockout mouse models, we have demonstrated that loss of Lpcat3 reduces membrane fluidity, impairs lipid metabolism in liver and intestine, and disrupts intestinal homeostasis. We are focusing on understanding how changes in membrane phospholipids and cholesterol contribute to human diseases using a multiple disciplinary approach that combines biochemistry, molecular and cell biology, genetically modified mouse models, and next generation sequencing.