Current Projects
Project: Identification of potential pathogenetic role of a copy number increase of the glycine decarboxylase gene in psychotic disorders.
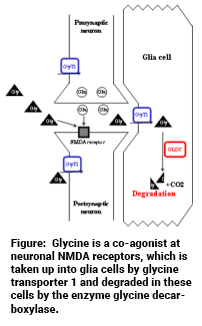
Structural variants like copy number variations (CNV), i.e., genomic deletions and duplications, have been discovered as rare mutations with sometimes large effect sizes in psychotic disorders. In a family affected by schizoaffective disorder and bipolar disorder with psychotic features, a small supernumerary marker chromosome containing genomic material from the 9p24.1 region was identified, that segregates with disease (J. TCW et al., Stem Cell Reports 2017;8:519-528; C.M. Grochowski et al., Human Mutation 2018;39:939-946). This results in a “duplication” (i.e., 3 copies instead of 2 copies) of some genes in this genomic region and a “triplication” (i.e., 4 copies instead of 2 copies) of the GLDC gene encoding glycine decarboxylase. Glycine decarboxylase degrade glycine, which is a co-agonist at the NMDA receptor. NMDA receptor hypofunction has been postulated to be a major pathophysiological factor in schizophrenia. Our working hypothesis is that the increased GLDC copy number results in increased amounts of the enzyme glycine decarboxylase, which then results in reduced glycine levels in astrocytes, from which it is known to be released by afferent innervation via AMPA receptors. This would then result in decreased availability of glycine at neuronal NMDA receptors and thus NMDA receptor hypofunction.
Using chromosome engineering (gene targeting followed by trans-chromosomal recombination), we have developed mice with 3 or 4 copies of the 9p24.1 genes, which demonstrate phenotypes consistent with neurodevelopmental disorders such as schizophrenia. We are also developing mice with increased copy numbers of only the Gldc gene and of the other 9p24.1 genes except Gldc. With these mice, we want to investigate whether the phenotypes caused by the large CNV mimicking the patients is due to copy number increase of Gldc or due to copy number increase of the other 9p24.1 genes. We want to study how these structural mutations affect biochemical risk pathways and behavior, and in collaboration with Dr. Vadim Bolshakov, an electrophysiologist at McLean Hospital and Harvard Medical School, how these mutations affect synaptic transmission and changes in intrinsic excitability of neurons.
Project: Dissection of hippocampal circuits mediating anxiety and fear
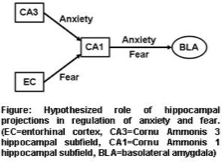 We have previously shown that while pharmacological reduction of anxiety (by the benzodiazepines diazepam) is dependent on α2-contianing GABAA receptors in dentate gyrus granule cells and CA3 pyramidal neurons, pharmacological reduction of fear is dependent on α2-contianing GABAA receptors in CA1 pyramidal neurons (E. Engin et al., eLife 2016;5:e14120; Engin et al., Trends in Pharmacological Sciences 2018;39:710-732). Our working hypothesis is that distinct intrahippocampal projections mediate anxiety and fear, with unique roles of the monosynaptic and the trisynaptic hippocampal pathways. We now want to determine how individual projections contribute to the bidirectional modulation of anxiety and fear using optogenetic approaches.
We have previously shown that while pharmacological reduction of anxiety (by the benzodiazepines diazepam) is dependent on α2-contianing GABAA receptors in dentate gyrus granule cells and CA3 pyramidal neurons, pharmacological reduction of fear is dependent on α2-contianing GABAA receptors in CA1 pyramidal neurons (E. Engin et al., eLife 2016;5:e14120; Engin et al., Trends in Pharmacological Sciences 2018;39:710-732). Our working hypothesis is that distinct intrahippocampal projections mediate anxiety and fear, with unique roles of the monosynaptic and the trisynaptic hippocampal pathways. We now want to determine how individual projections contribute to the bidirectional modulation of anxiety and fear using optogenetic approaches.
Project: Cellular mechanisms for age-related cognitive dysfunction and its pharmacological reversal: a strategy towards prevention and treatment of postoperative cognitive deficits
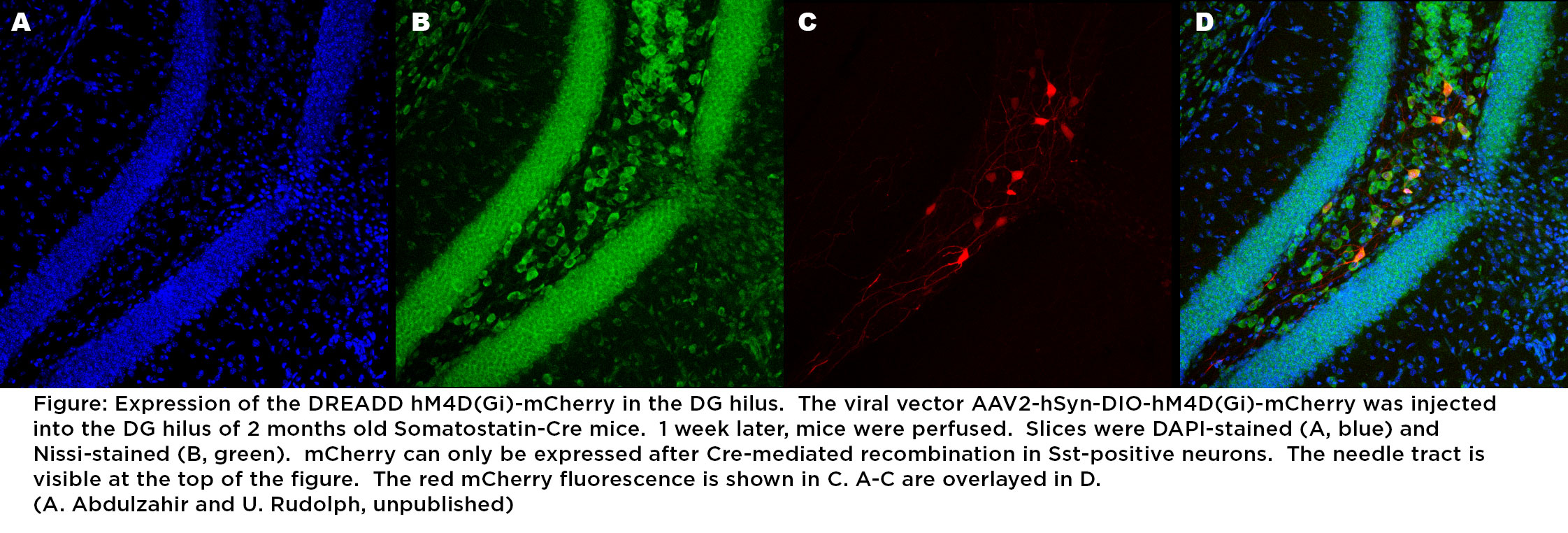
Learning and memory functions in the hippocampus are under the modulatory control of α5-containing GABAA receptors (E. Engin et al., Journal of Neuroscience 2015;35:13698-13712; E. Engin et al., Trends in Pharmacological Sciences 2018;39:710-732). They are also known to be critical mediators of the amnestic effects of the general anesthetic etomidate. Postoperative cognitive dysfunction is much more frequent in elderly patients than in young patients, indicating that age-related changes may underlie this increased propensity. Using chemogenetic methods, we want to investigate whether age-related changes that have been observed in rodents such as a reduced function and number of somatostatin-positive interneurons in the dentate gyrus hilus are the pathophysiological basis of age-related cognitive dysfunction. We further want to investigate whether a cognition-enhancing effect of intermittent doses of general anesthetics is dependent on α5-containing GABAA receptors, and whether a pharmacologically induced increase in the expression of α5-containing GABAA receptors can prevent or ameliorate postoperative cognitive dysfunction in aged mice. The project is expected to provide insights into molecular and cellular mechanisms underlying postoperative cognitive impairments and their reversal in aged mice. This project will be pursued in collaboration with Robert A. Pearce, Department of Anesthesiology, University of Wisconsin – Madison.