Conversion of an Esr1+ cell to Esr2+ cell during ovarian development
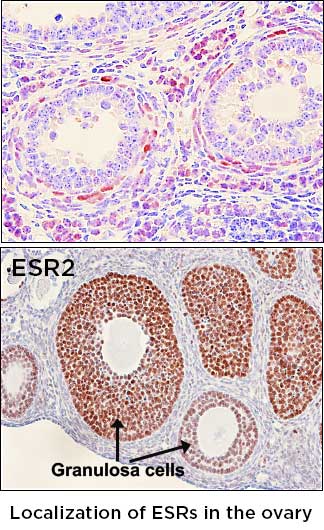 17β-estradiol (E2) is a steroid hormone that regulates a plethora of reproductive, metabolic, immunologic, cognitive, and skeletal functions. Two classical estrogen receptors, ESR1 (ERα) and ESR2 (ERβ) are responsible for the classical actions of E2 in mammalian species. In the ovary, ESR1 is localized to the surface epithelium and theca cells, whereas ESR2 is expressed in the granulosa cells. In previous studies, we have generated a transgenic mouse line that has an insertion of an enhanced Cre (iCre) recombinase in the exons of Esr1 and Esr2 gene, respectively. Esr1Cre mice can be used for deleting any floxed gene in the ESR1-expressing (Esr1+) cells, and Esr2Cre mice for excising genes in ESR2-expressing (Esr2+) cells. These novel transgenic mice are being used for lineage-tracing Esr1+ and Esr2+ cells and investigating unique roles that these estrogen receptors play in a single lineage. One interesting finding that we made is that Esr2+ granulosa cell seems to originate from cells that once expressed ESR1. I hypothesize that there is a molecular switch that convers Esr1+ cells to Esr2+ cells in developing ovary. In this project, 1) I will determine the temporal window when ESR1/ESR2 switching occurs, 2) the role of oocyte in granulosa cell lineage determination and 3) the mechanism of silencing Esr1 promoter during the transition of ESR1 to ESR2 granulosa cells. A discovery of the existence of a mechanism that converts ERa+ cells to ERb+ cells is expected to open-up a possibility of changing Esr1+ cells to Esr2+ cells.
17β-estradiol (E2) is a steroid hormone that regulates a plethora of reproductive, metabolic, immunologic, cognitive, and skeletal functions. Two classical estrogen receptors, ESR1 (ERα) and ESR2 (ERβ) are responsible for the classical actions of E2 in mammalian species. In the ovary, ESR1 is localized to the surface epithelium and theca cells, whereas ESR2 is expressed in the granulosa cells. In previous studies, we have generated a transgenic mouse line that has an insertion of an enhanced Cre (iCre) recombinase in the exons of Esr1 and Esr2 gene, respectively. Esr1Cre mice can be used for deleting any floxed gene in the ESR1-expressing (Esr1+) cells, and Esr2Cre mice for excising genes in ESR2-expressing (Esr2+) cells. These novel transgenic mice are being used for lineage-tracing Esr1+ and Esr2+ cells and investigating unique roles that these estrogen receptors play in a single lineage. One interesting finding that we made is that Esr2+ granulosa cell seems to originate from cells that once expressed ESR1. I hypothesize that there is a molecular switch that convers Esr1+ cells to Esr2+ cells in developing ovary. In this project, 1) I will determine the temporal window when ESR1/ESR2 switching occurs, 2) the role of oocyte in granulosa cell lineage determination and 3) the mechanism of silencing Esr1 promoter during the transition of ESR1 to ESR2 granulosa cells. A discovery of the existence of a mechanism that converts ERa+ cells to ERb+ cells is expected to open-up a possibility of changing Esr1+ cells to Esr2+ cells.
Dynamic Shift of ESR2 to ESR1 Expression in the Female Reproductive Organs
Estrogen, primarily through its receptors ESR1 and ESR2, plays a critical role in reproductive development, function, and systemic physiology. Traditionally, these receptors were thought to function independently within distinct cell types; however, recent evidence suggests a dynamic switch in receptor expression between ESR1 and ESR2 is essential for maintaining reproductive health. This proposal investigates the physiological and pathological consequences of ESR2-to-ESR1 receptor switching in the hypothalamic-pituitary-ovarian (HPO) axis and uterus using innovative transgenic mouse models, including Esr2-Esr1KO and Esr1-Esr2KO mice. The project focuses on understanding how this receptor switching impacts neuroendocrine regulation, ovarian function, and uterine pathology, including inflammation and hyperplasia.
The proposed studies will use advanced methodologies, including single-cell RNA sequencing, immunohistochemistry, lineage tracing, and functional assays, to identify the spatial and temporal dynamics of receptor switching and its contribution to reproductive phenotypes. This work will address critical gaps in our understanding of estrogen receptor biology, uncover novel mechanisms underlying infertility and estrogen-related uterine disorders, and pave the way for innovative therapeutic approaches in women’s health.
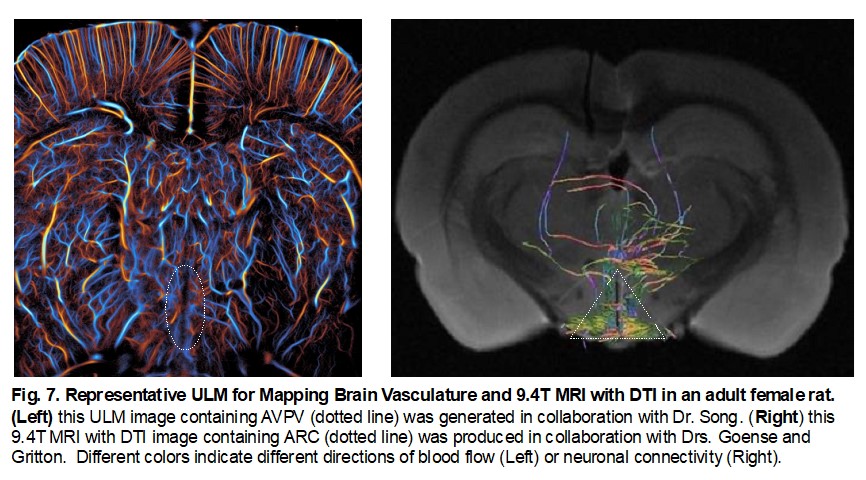
In Vivo Imaging of Neurovascular and Neuro-connectivity Changes in the Hypothalamus Induced by Neonatal Estrogen Exposure
The hypothalamus plays a central role in regulating critical biological processes such as reproduction, metabolism, and homeostasis. However, current methods for studying hypothalamic function are limited to postmortem analyses. This study aims to develop and validate a non-invasive imaging framework combining high-resolution 9.4T MRI, diffusion tensor imaging (DTI), and ultrasound localization microscopy (ULM) to detect and quantify neuroanatomical and vascular changes in the hypothalamus. Using a neonatally estrogen-exposed rat model, this research will provide real-time insights into estrogen-induced changes in hypothalamic structure and function. These advancements will enhance our understanding of neuroendocrine regulation and offer a new diagnostic tool for disorders related to hypothalamic dysfunction, such as infertility and neurodevelopmental conditions.
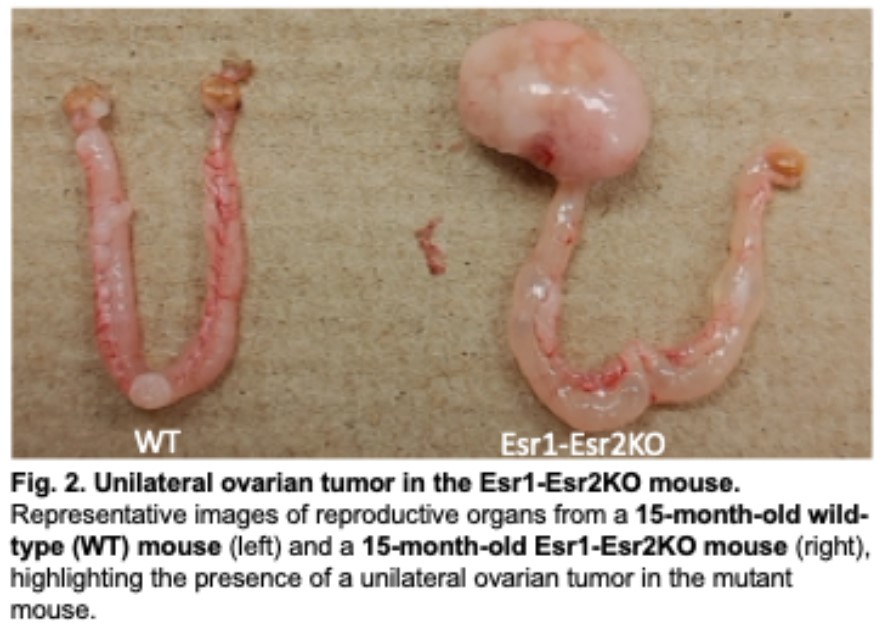
Adult-type Granulosa Cell Tumor – Tumorigenesis, Serum biomarkers and Treatment
Adult-type granulosa cell tumor (AGCT) is a rare ovarian cancer that accounts for approximately 2–5% of all ovarian malignancies and is typically diagnosed around 50 years of age. Although AGCTs are slow-growing, they exhibit a high rate of recurrence and currently lack effective biomarkers for early detection or therapeutic monitoring. Over 97% of AGCTs harbor a somatic FOXL2^C134W mutation, yet the initiating events, tumor-promoting microenvironmental factors, and downstream molecular mechanisms remain largely undefined.
This proposal aims to uncover the mechanisms underlying AGCT initiation and progression using two newly developed and complementary mouse models: (1) GC-Foxl2C130W, which expresses the pathogenic FOXL2 mutation specifically in granulosa cells and recapitulates human AGCT features, and (2) Esr1-Esr2KO, a novel model that exhibits aberrant estrogen receptor switching and spontaneous AGCT-like tumors with high phenotypic fidelity to the human disease. Remarkably, Esr1-Esr2KO tumors exhibit elevated serum estradiol, presence of Call-Exner bodies, and unilateral tumor formation, offering a robust and reproducible platform for mechanistic investigation.
This research builds on our laboratory’s longstanding expertise in reproductive endocrinology, estrogen receptor signaling, and transgenic modeling. By integrating high-resolution molecular profiling with functional and translational studies, this project will clarify the pathogenesis of AGCT, discover early biomarkers, and establish preclinical evidence for targeted therapies. The proposed work addresses a significant unmet need in ovarian cancer research and will lay the groundwork for improved diagnostic and therapeutic strategies for AGCT patients.