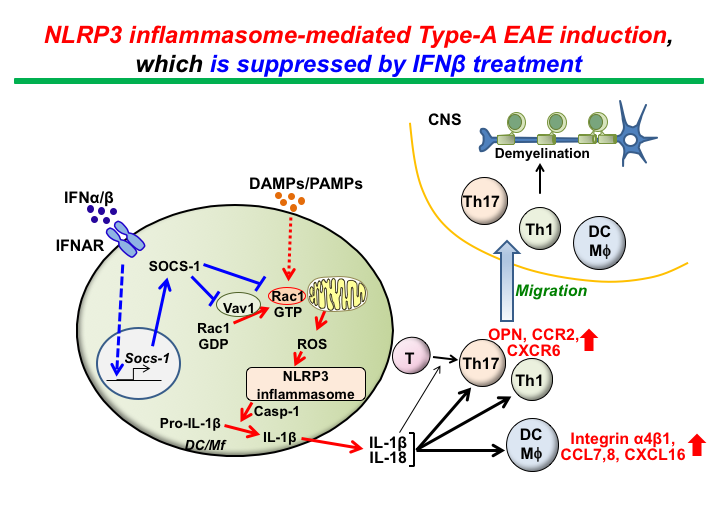
We study molecular mechanisms that induce heterogeneity in multiple sclerosis (MS) pathology in an effort to provide new molecular targets for the identification of biomarkers able to predict treatment outcome of therapy with interferon β (IFNβ), a well-known, first-line treatment of MS. As such, this may allow development of new personalized treatments for MS patients. We have demonstrated that NLRP3 inflammasome is a pathogenic factor in conventional types of experimental autoimmune encephalitis (termed Type-A EAE). Type-A EAE occurs by NLRP3 inflammasome-induced enhanced cell recruitment within the central nervous system (CNS) rather than via enhancing Th17 responses (PNAS 2012). Furthermore, we have found that IFNβ suppresses NLRP3 inflammasome activity via previously uncharacterized signaling pathways (Sci.Signal. 2012). In contrast, NLRP3-inflammasome-independent EAE (termed Type-B EAE) can thus be induced by using a system commensurable to Type-A EAE induction (Nat. Neurosci. 2016). Importantly, the Type-A vs. Type-B EAE models allowed study of the pathology of disease heterogeneity. More recently, we have discovered that Type-B EAE shows prolonged disease phenotype with irreversible neuronal loss via semaphoring 6B. In addition, Type-B EAE cannot be treated with IFN-β, a first-line drug to treat MS. Membrane-bound lymphotoxin-β receptor (LTβR) and CXCR2 are involved in Type-B EAE development, and Type-B EAE is ameliorated by antagonizing the receptors. Phenotypes of Type-A and Type-B EAE may be reminiscent of relapsing-remitting MS and progressive MS, respectively. This study will be further addressed in future research.
This study was done at Duke University under Dr. Shinohara’s supervision.