ONGOING RESEARCH
Mouse Projects
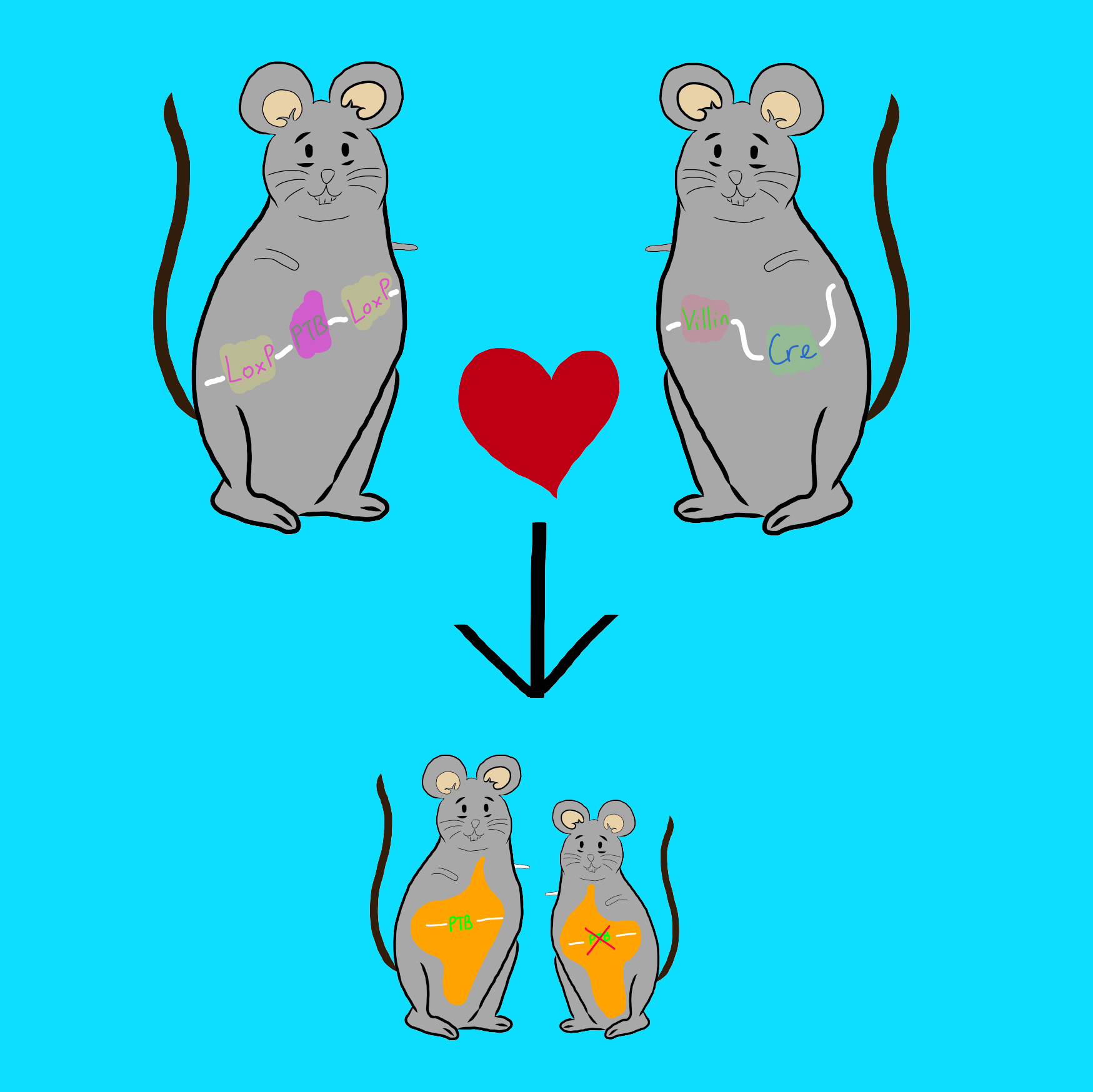
We recently generated a mouse model in which hnRNP I, a master regulator of post-transcriptional regulation, is disrupted in mouse intestinal epithelial cells. This results in impaired host-microbe interactions in the neonatal stage followed by spontaneous colitis and early onset of invasive colorectal cancer. These defects, which link impaired neonatal host-microbe interactions to development of colitis and colorectal cancer, are not found in other murine models of colitis-associated colorectal cancer. Currently, we use the hnRNP I knockout mouse model as a tool to study the mechanism that is important for establishing a mutualistic relationship between the host and gut microbiota in the neonatal stage. Moreover, we use this mouse model to identify the alterations of gut microbiota that are associated with the risk of developing colitis-associated colorectal cancer, and to determine their contribution to colorectal carcinogenesis. Lastly, we use this mouse model to determine how beneficial microbes modulate the intestinal microbiota and host intestinal immunity to prevent colitis-associated colorectal cancer.
To determine the age-specific function of hnRNP I in intestinal epithelial cells, we generated a tamoxifen-inducible hnRNP I knockout mouse model. We are currently investigating the role of hnRNP I in regulating intestinal stem cell survival in adulthood.

Zebrafish Projects
We use zebrafish as an alternative model to study aspects of host-microbe interactions that are difficult to be assessed in mice. Zebrafish and mice share conserved host gene response to their compositionally distinct microbial communities. Compared to mice, zebrafish provide the opportunity to assess large populations of hosts and to visualize microbe-host associations at a cellular level in living animals. Generation of germ-free zebrafish is relative easy and cost effective. Specifically, we are interested to use zebrafish to study the impact of potentially disease-promoting microbes identified in colitis and colorectal cancer patients on the host immunity. We are also interested to study how beneficial microbes impact host health through generating health-promoting metabolic products.