By Alex Gochenauer, PharmD, DICVP, FSVHP
With the development of more complex drugs, confusion has arisen regarding the difference between biosimilars and generics.
A biosimilar is a biologic molecule that is almost identical to an FDA-approved “original” biologic medication but is manufactured by a different company. Since biologics come from living organisms, there is no “recipe” for their development, unlike standard medications.
Generic medications are developed to be the exact same as an already marketed brand-name drug in dosage form, safety, strength, route of administration, quality, performance characteristics, and intended use.
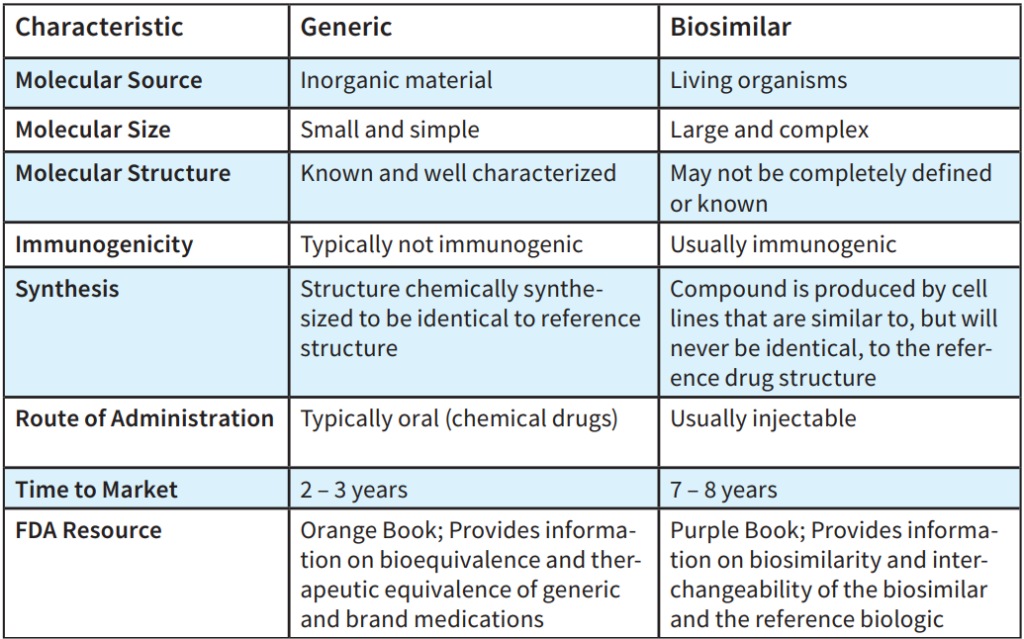
The table above summarizes key differences between generic medications and biosimilars.
An example of a generic would be utilizing the branded product Rimadyl (carprofen) as a reference and developing a generic carprofen product that is bioequivalent. An example of a biosimilar is utilizing Lantus (insulin glargine) as a reference and developing the biologic Semglee (insulin glargine-yfgn). Semglee contains a slightly different form of insulin glargine, but this does not lead to any clinically meaningful difference between the two products.
Don’t make the mistake of thinking Basaglar (insulin glargine) is a biosimilar for Lantus: it is considered a follow-on biologic and is not interchangeable with Lantus or Semglee, as it received approval before insulin was required to go through the biosimilar approval pathway.




